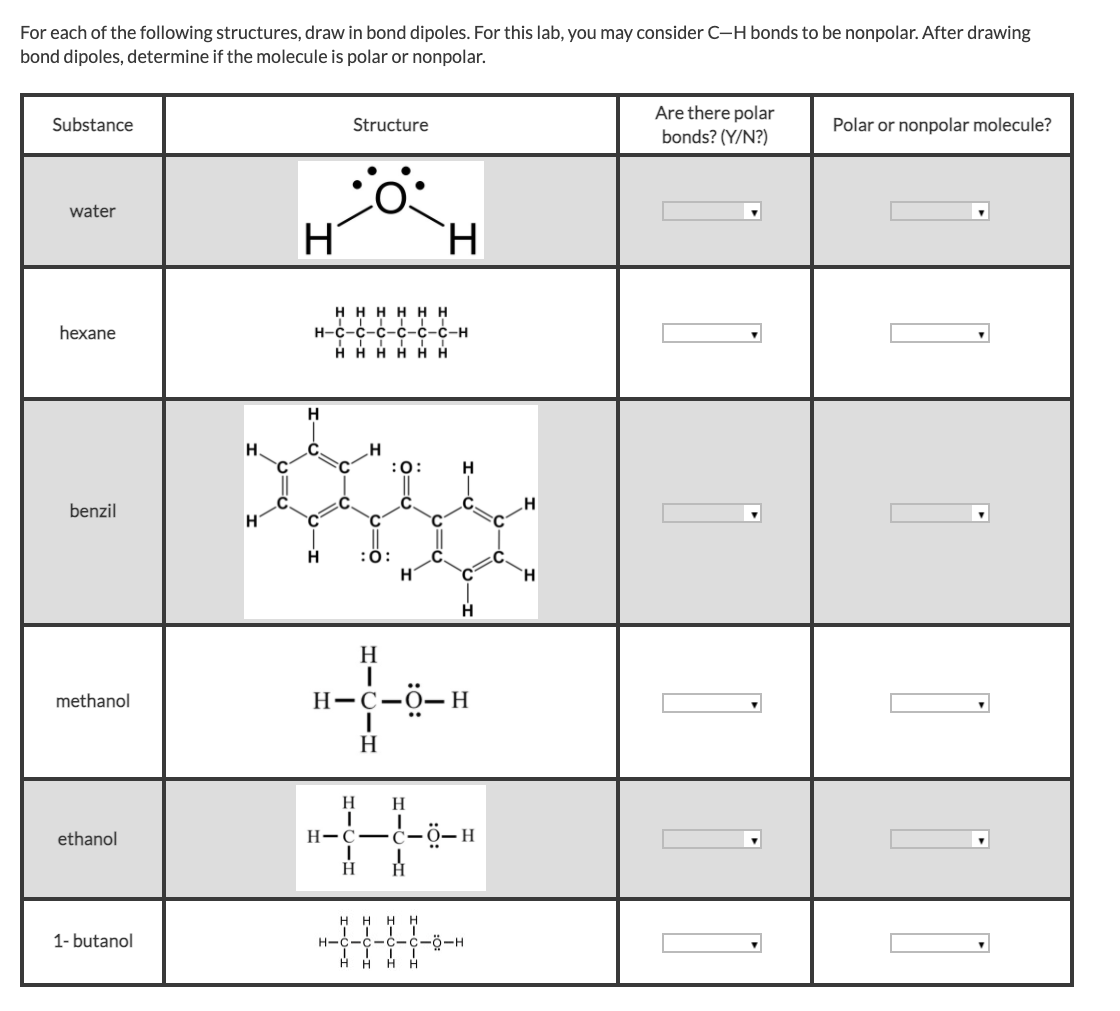
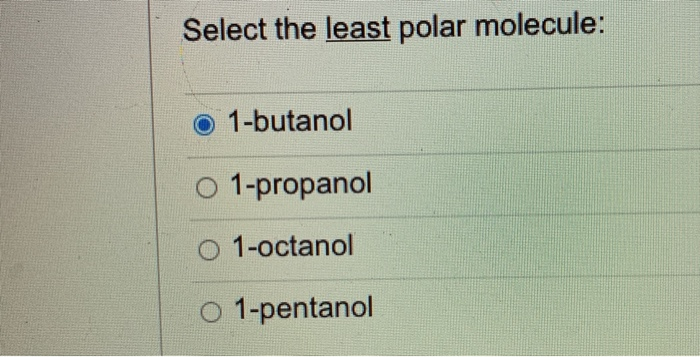
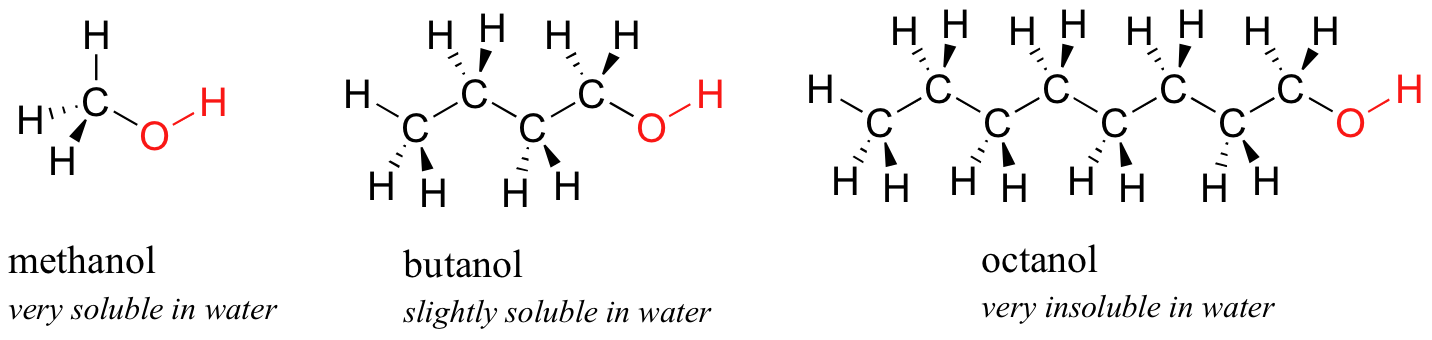
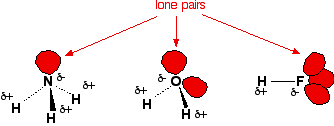
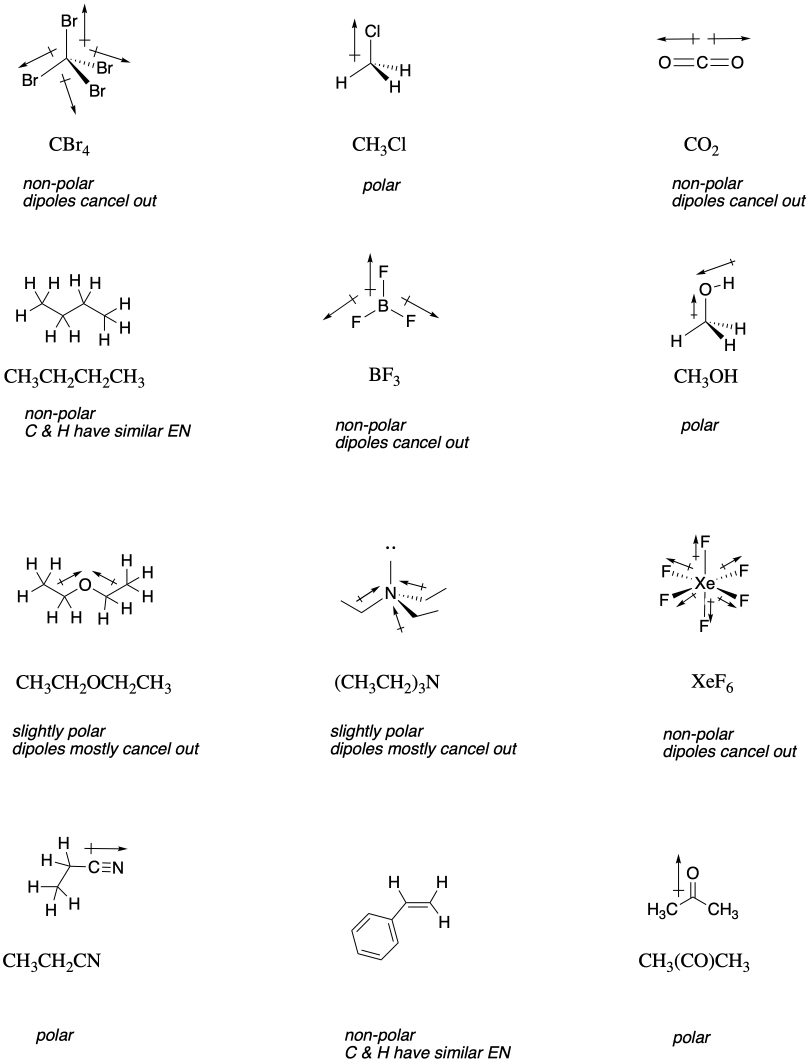
Which of these would have the greatest polarity? I know its one of the propanols but which one? : r/chemhelp

Is 1-butanol an ionic, molecular nonpolar, or molecular polar compound? What intermolecular forces are present? | Homework.Study.com

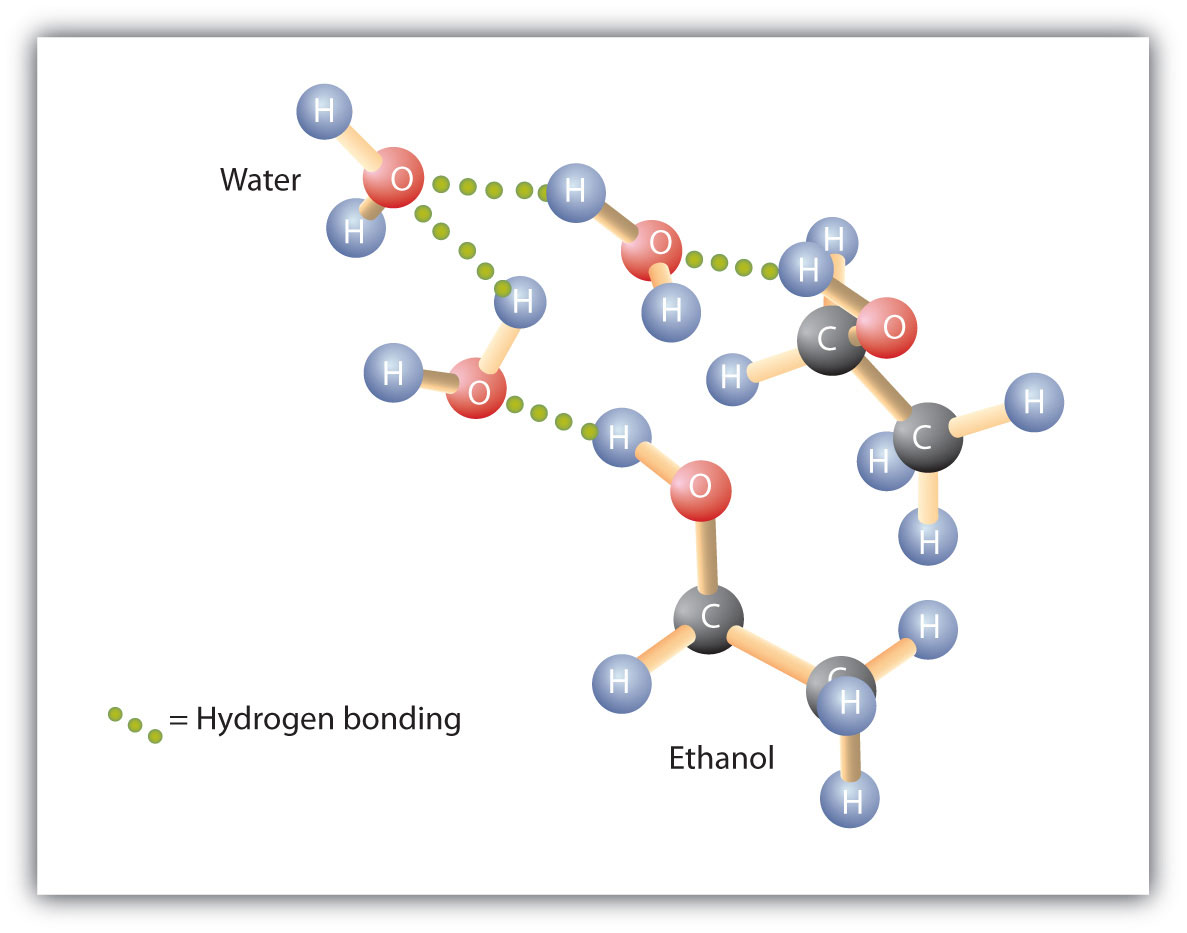
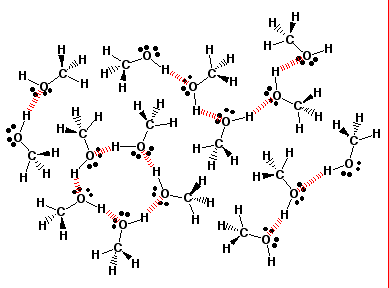
1-Butanol as a Solvent for Efficient Extraction of Polar Compounds from Aqueous Medium: Theoretical and Practical Aspects | The Journal of Physical Chemistry B