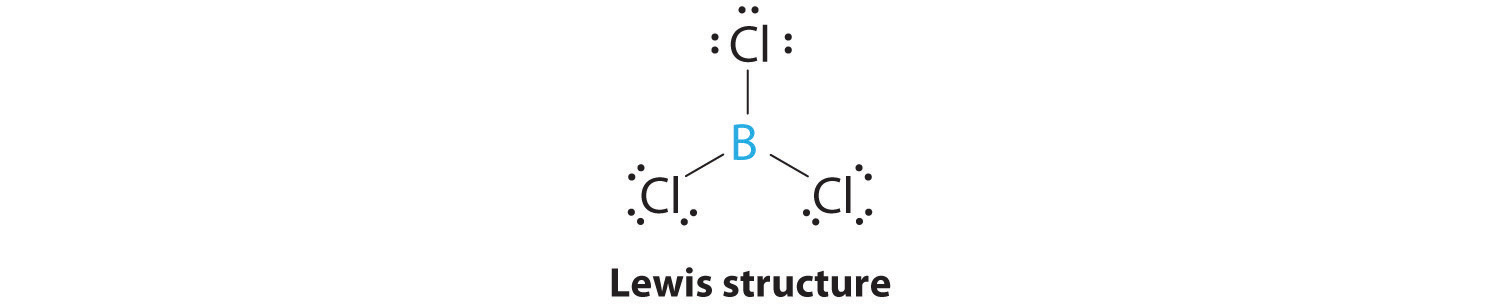
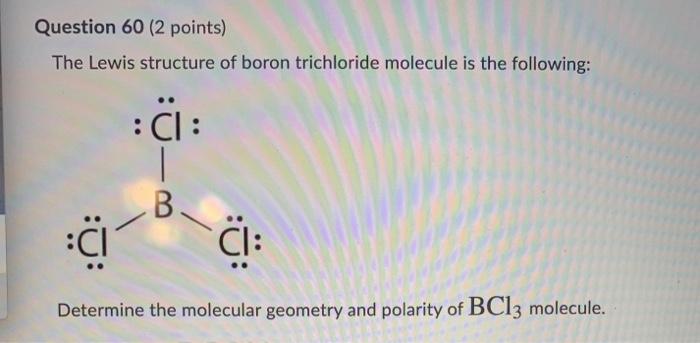
Consider the following statements about BCl3 molecules. Which statement is false? A. The B-Cl bonds are quite polar. B. The bond dipoles exactly cancel. C. The Cl-B-Cl bond angles are 109.5 degrees.

BCl3 Lewis Structure (Boron Trichloride) | BCl3 Lewis Structure (Boron Trichloride) Welcome back to our channel and in today's video we will help you determine the Lewis Structure of Boron... | By

BCl3 Lewis Structure: How to Draw the Dot Structure for BCl3 | Chemical Bonding | Success in Chemistry

Why is bcl3 trigonal planar in shape whereas anhydrous alcl3 is tetrahedral in shape - Chemistry - Chemical Bonding and Molecular Structure - 12837253 | Meritnation.com

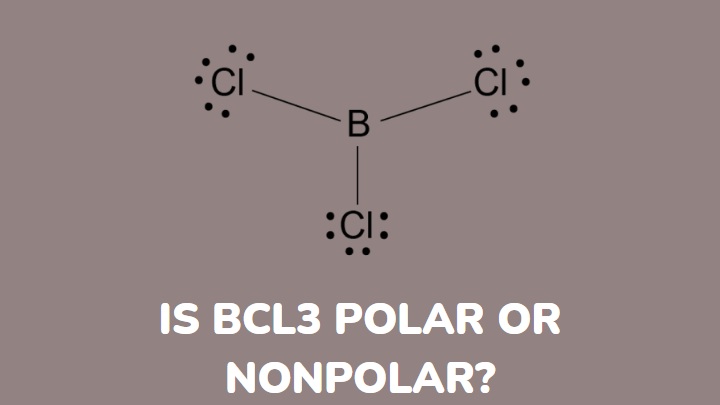
Is BCl3 Polar or Non-polar? (Boron Trichloride) | Is BCl3 Polar or Non-polar? (Boron Trichloride) Were you searching for a video that can help you find out if BCl3? If yes then