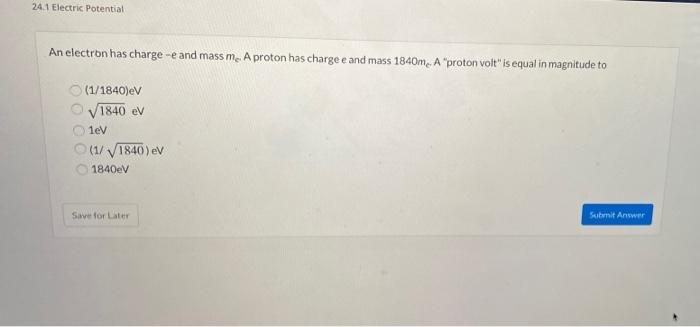
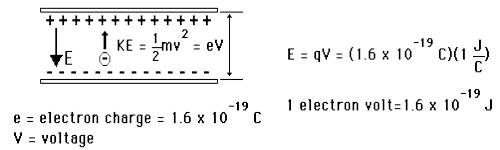
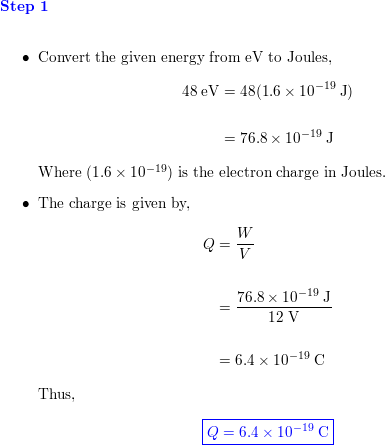An electron is accelerated through voltage. Its frequency will be (e = charge on electron, h = Planck's constant).

SOLVED: Electron Proton Calculate the potential energy of an electron and proton in a hydrogen atom (use 1 eV = 1.6 x 10^-19 J): In Joules: In eV: Use the fact that

77. An electron of 100 eV is fired directly towards a metal plate having surface charge density of -2 x 106 Cm 2. What is the distance from where the electron be

The electric potential energy of electron-proton system of hydrogen atom is: (Given : The radius of electron orbit = 0.53overset {circ}{A}, electronic charge = 1.6times 10^{-19}C).

A single electron ion has nuclear charge + Ze where Z is atomic number and e is electronic charge. It requires 16.52 eV to excite the electron from the second Bohr orbit