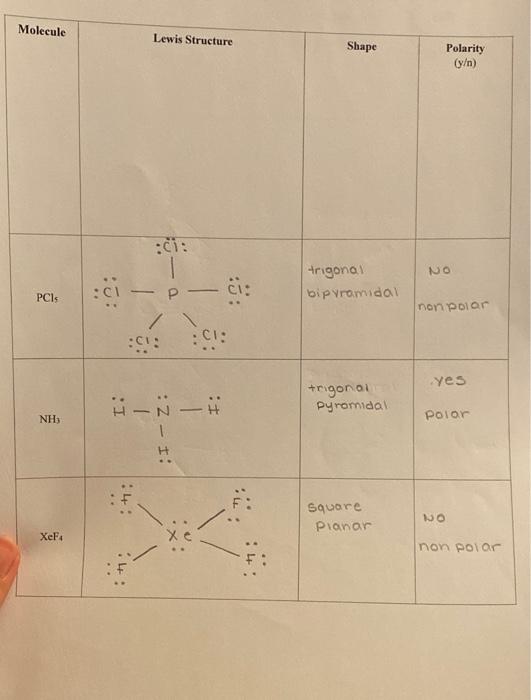
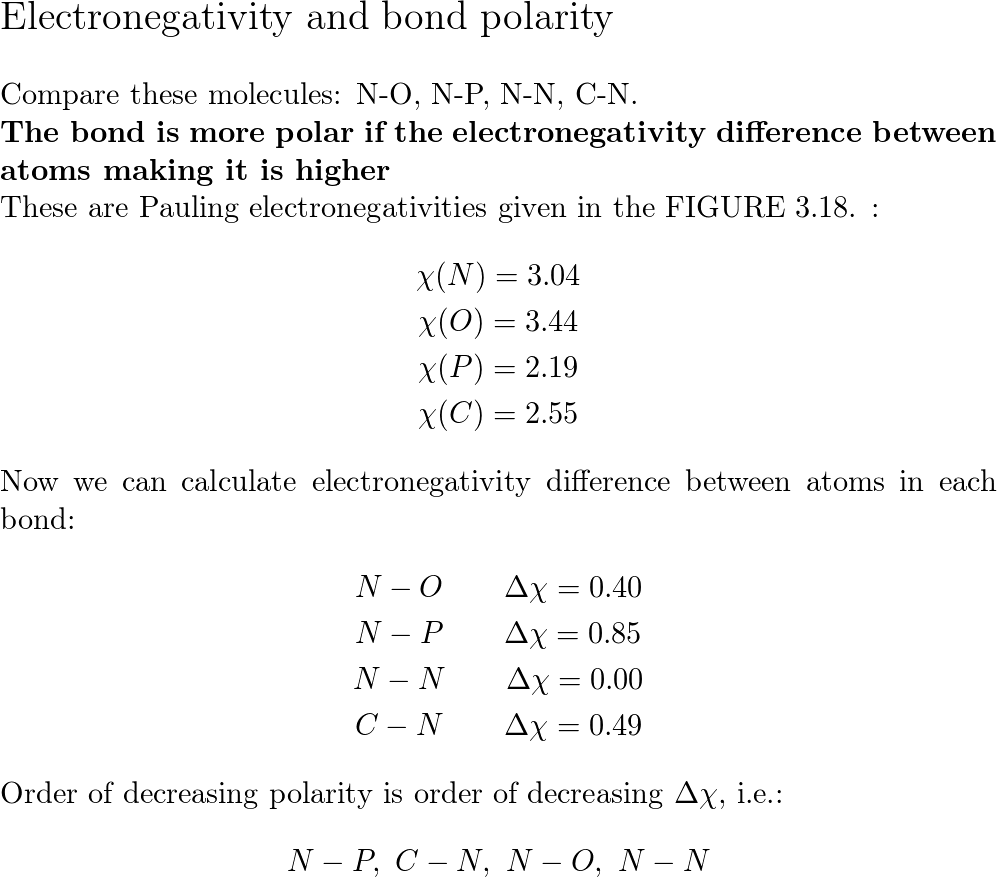Determine the net dipoles in a molecule of HCN. Indicate whether or not the molecule will be polar. | Homework.Study.com

Evidence for Polarity- and Viscosity-Controlled Pathways in the Termination Reaction in the Radical Polymerization of Acrylonitrile | Macromolecules

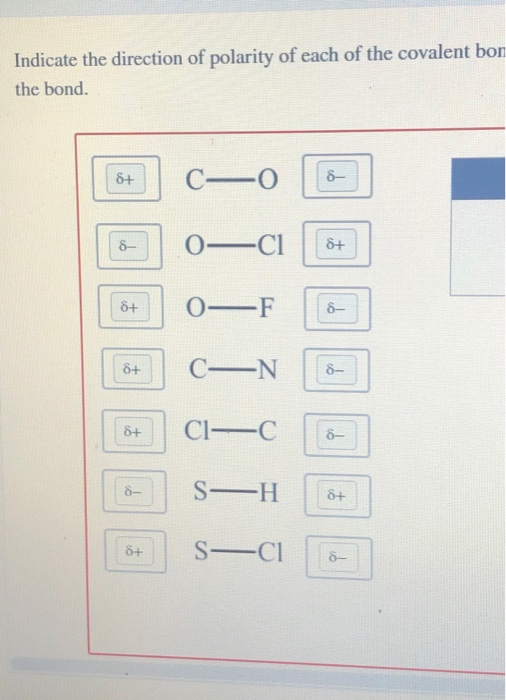
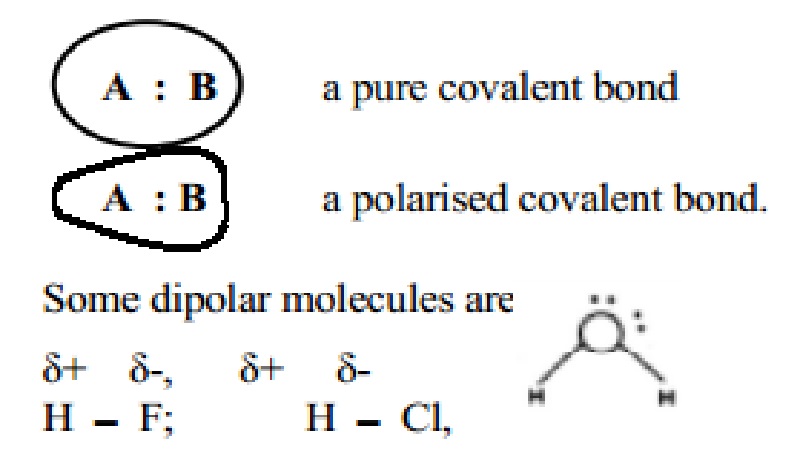
Indicate the direction of polarity of each of the covalent bonds by placing the appropriate delta notation - brainly.com

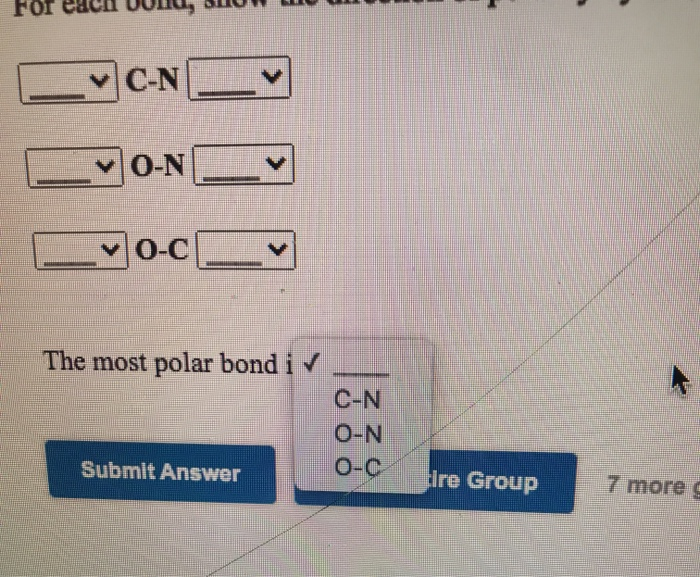
For each pair of bonds, indicate the more polar bond, and use an arrow to show the direction of polarity in - brainly.com

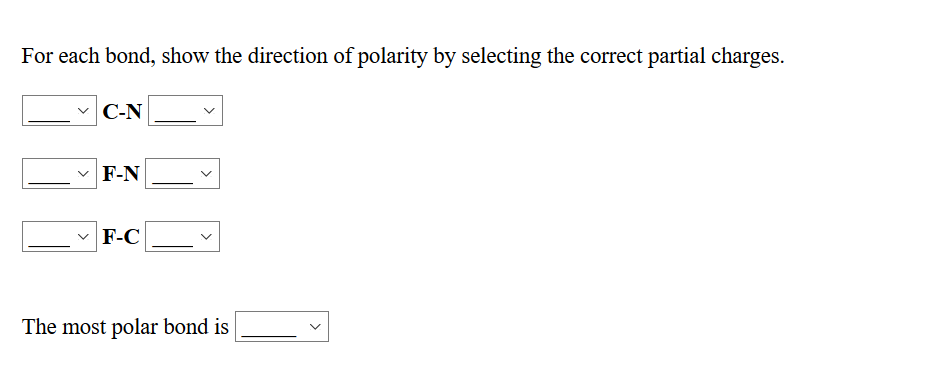
Using the symbols delta- and delta+, indicate the direction of polarity in each polar covalent bond. (a) C-N (b) N-O (c) C-Cl | Homework.Study.com

Design strategies for high‐polarity host materials and their chemical... | Download Scientific Diagram