Intriguing Electrostatic Potential of CO: Negative Bond-ends and Positive Bond-cylindrical-surface | Scientific Reports

D. Which of the following is true CO,? C=0 bond Co, molecule polar non-polar non-polar polar polar polar D non-polar non-polar
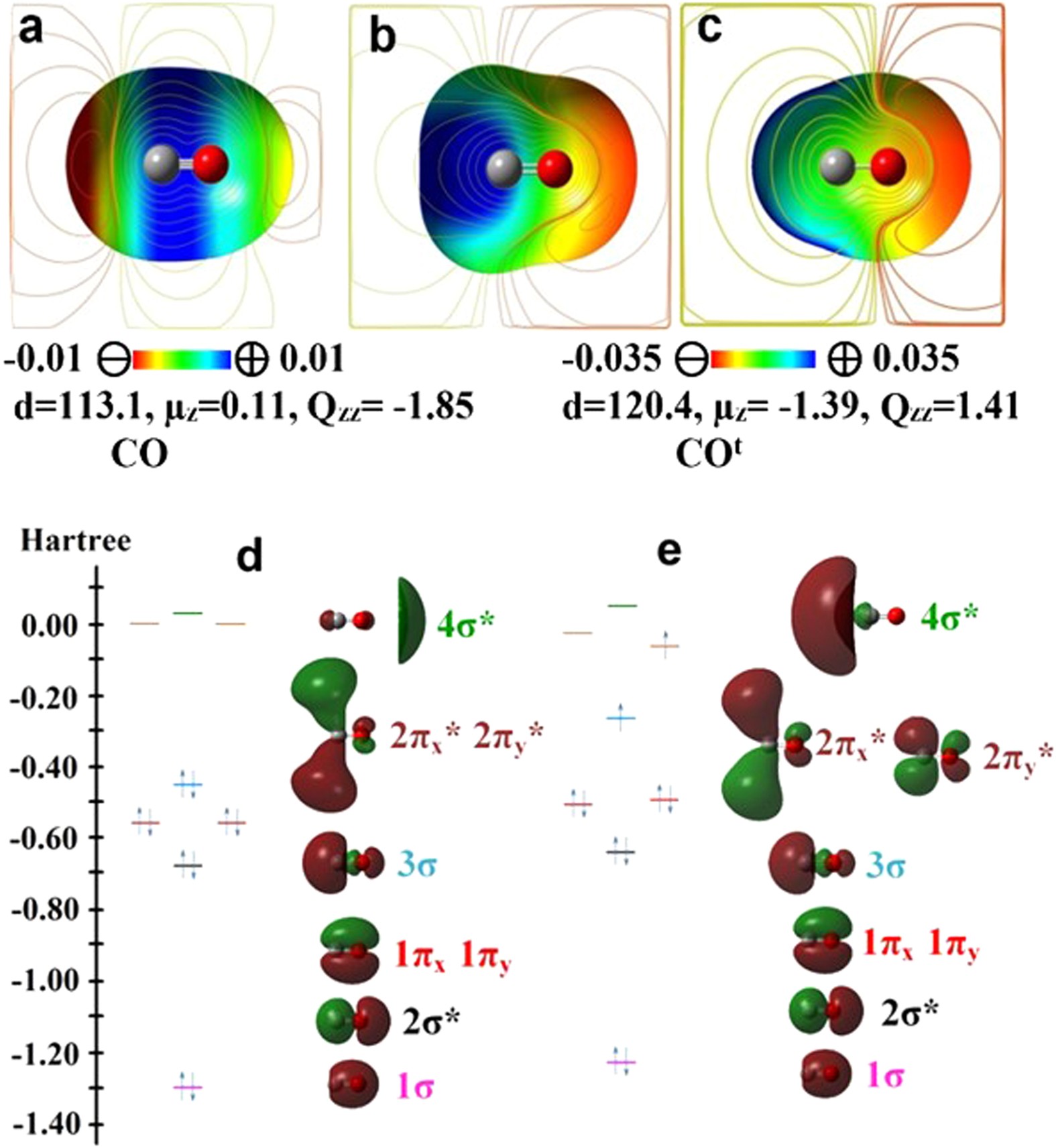
Intriguing Electrostatic Potential of CO: Negative Bond-ends and Positive Bond-cylindrical-surface | Scientific Reports





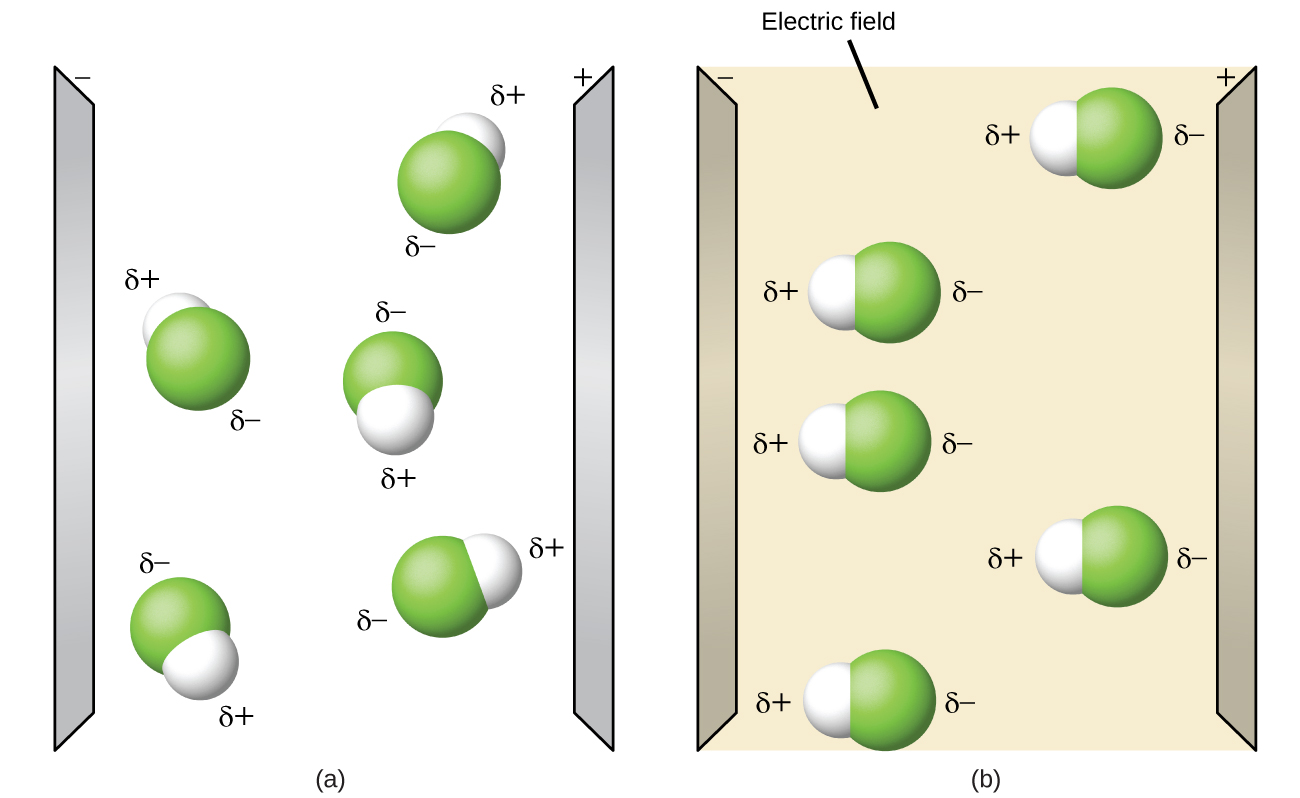

![Bengali] CO(2) is non-polar but COS is polar, even though both of the Bengali] CO(2) is non-polar but COS is polar, even though both of the](https://static.doubtnut.com/ss/web-overlay-thumb/4044529.webp)