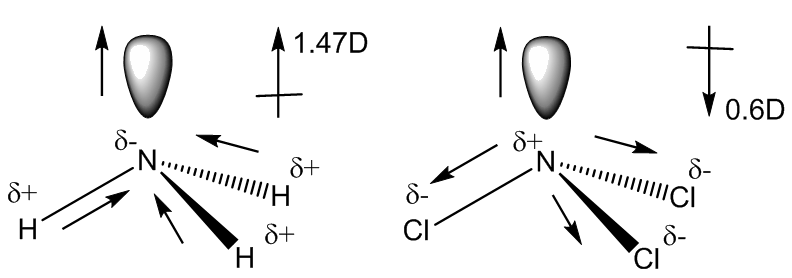
Of the molecules listed, which does not have a dipole moment? a. HCl b. NCl3 c. CO d. BF3 e. All molecules have a dipole moment. | Homework.Study.com

Are molecules of the following compounds polar or nonpolar? a) HF b) CS2 c) CH4 d) NCl3 | Homework.Study.com