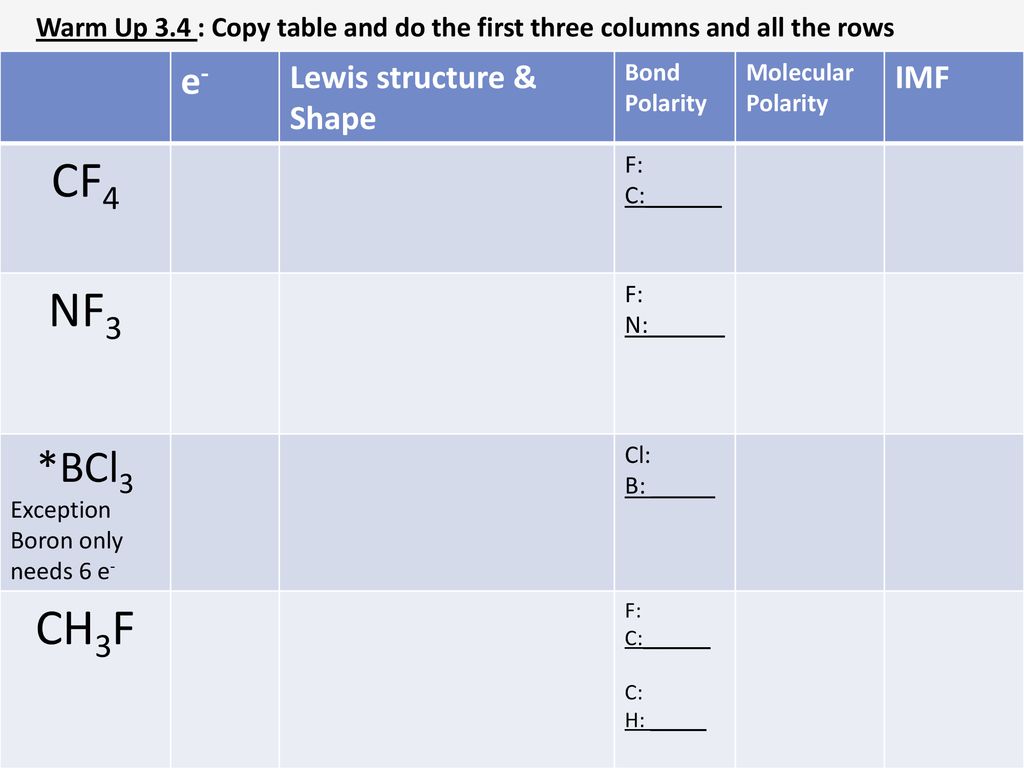
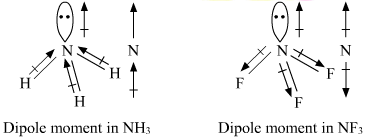
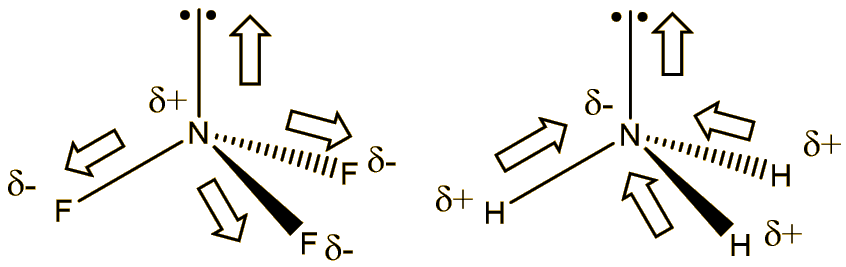
BF_{3} and NF_{3} both are covalent compounds but NF_{3} is polar whereas BF_{3} is non-polar. This is because :

Is NF3 Polar or Nonpolar? - Polarity of Nitrogen trifluoride | Molecular geometry, Polar, Covalent bonding

In a polar nitrogen trifluoride, NF3 molecule, nitrogen and fluorine atoms share electrons. The fluorine - brainly.com

Which statement for NH3 and NF3 is false? Electro negativities N = 3.0, H = 2.1, F = 4.0 - Home Work Help - Learn CBSE Forum

write lewis structures of nf3. what is the electronic and molecular geometry? is the molecule polar or - brainly.com





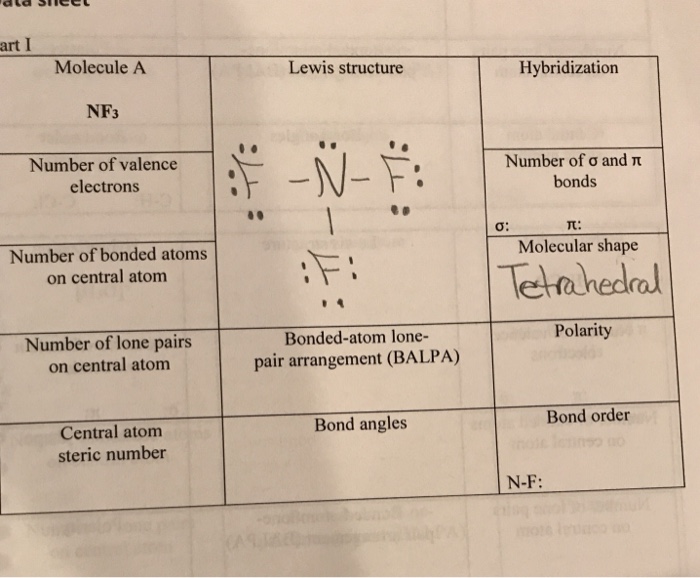



![ANSWERED] A Is CH O polar or nonpolar B Is NF3 polar... - Organic Chemistry - Kunduz ANSWERED] A Is CH O polar or nonpolar B Is NF3 polar... - Organic Chemistry - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20220502230749422436-4415097.jpg)