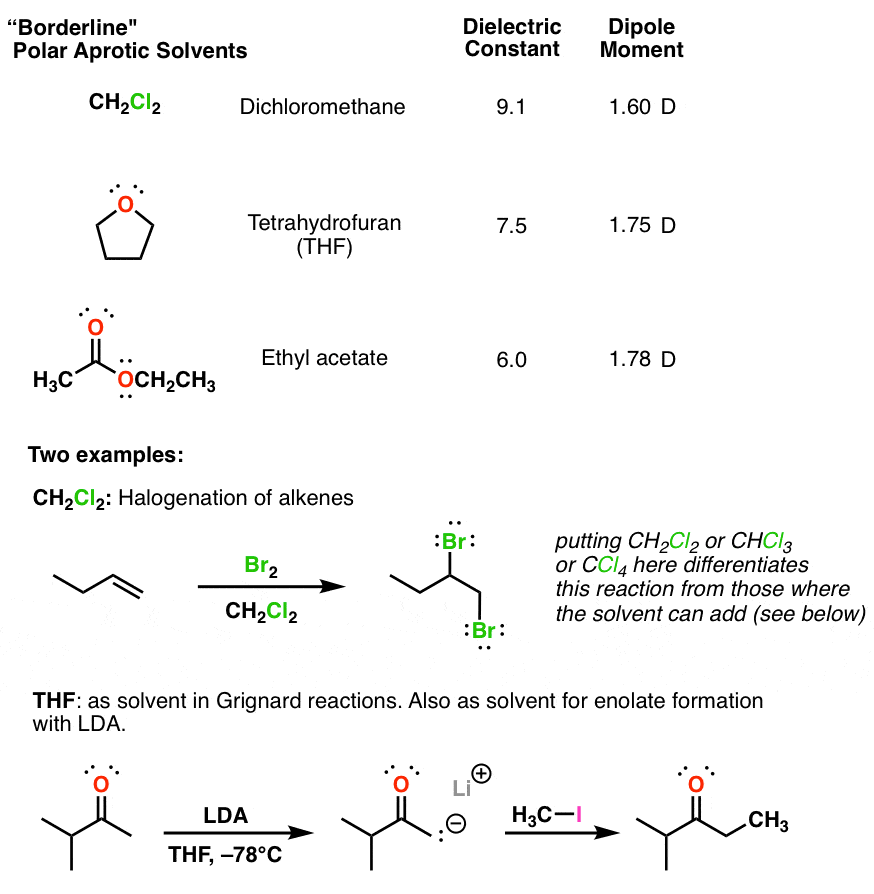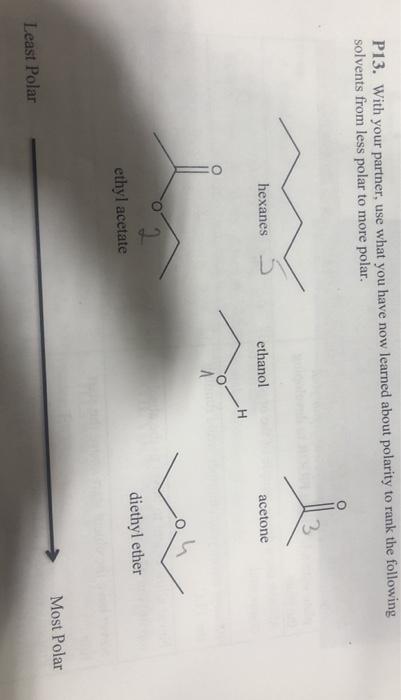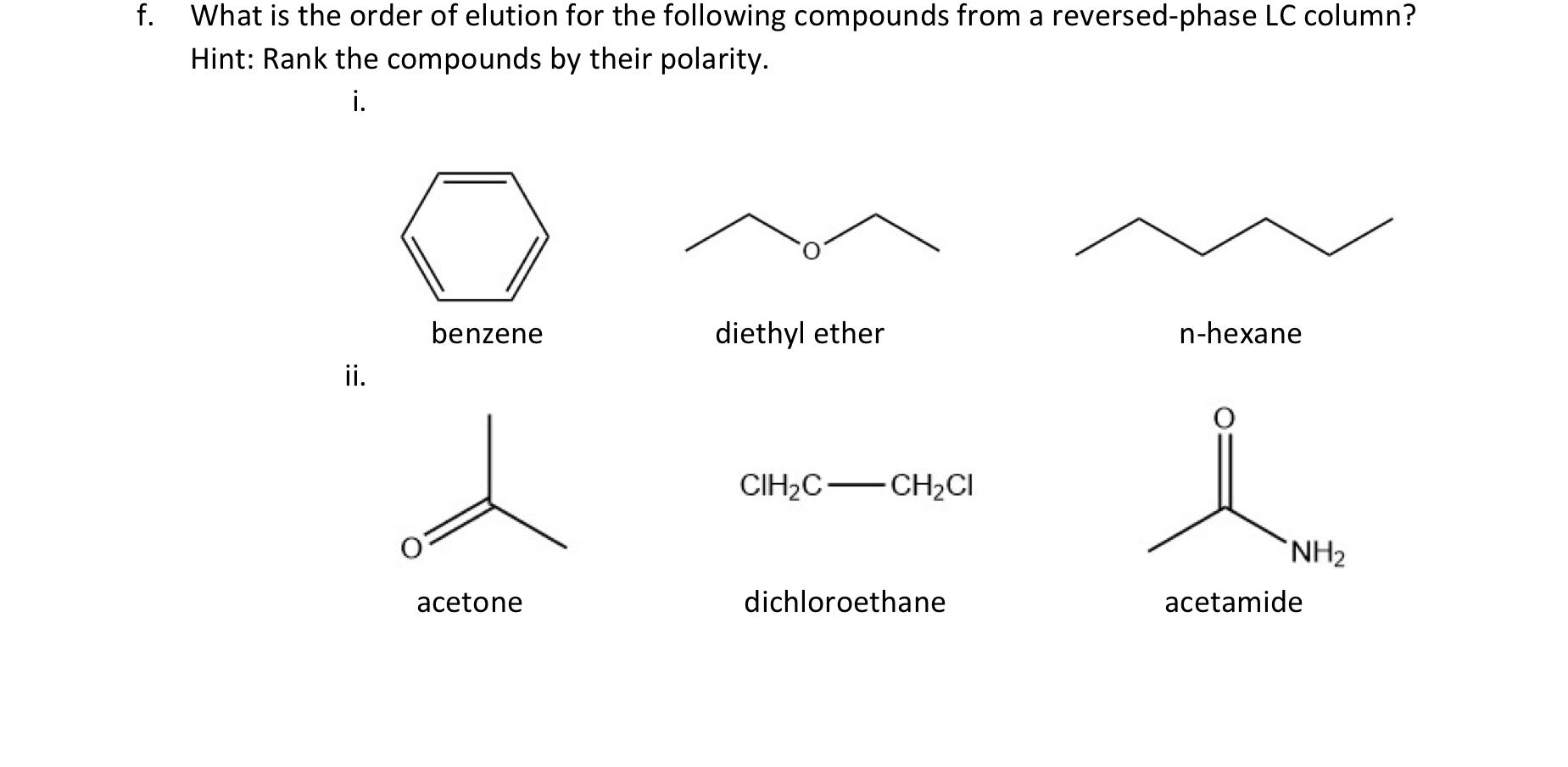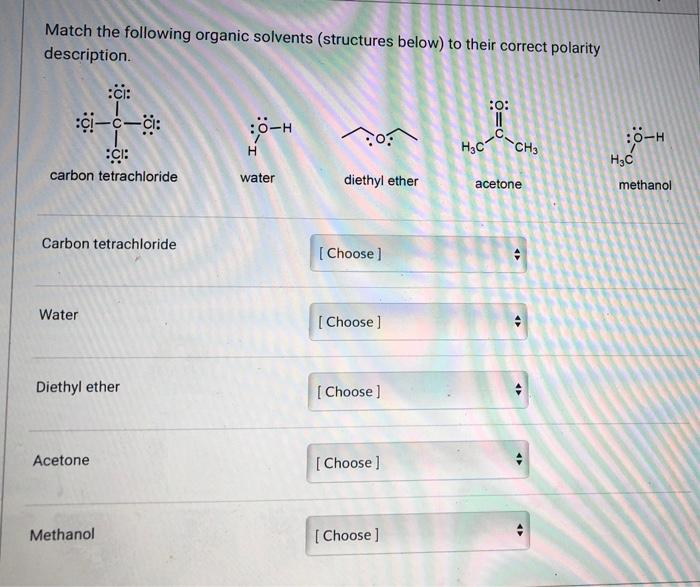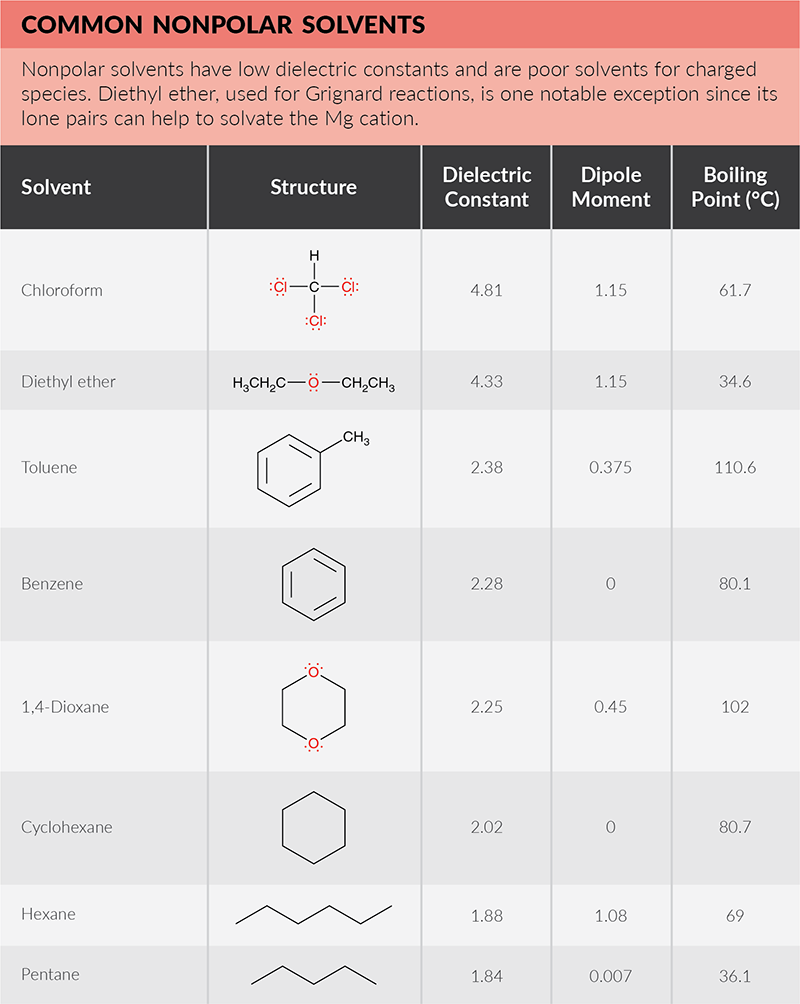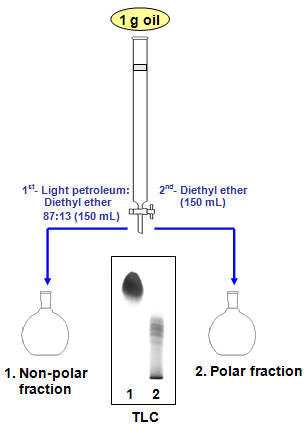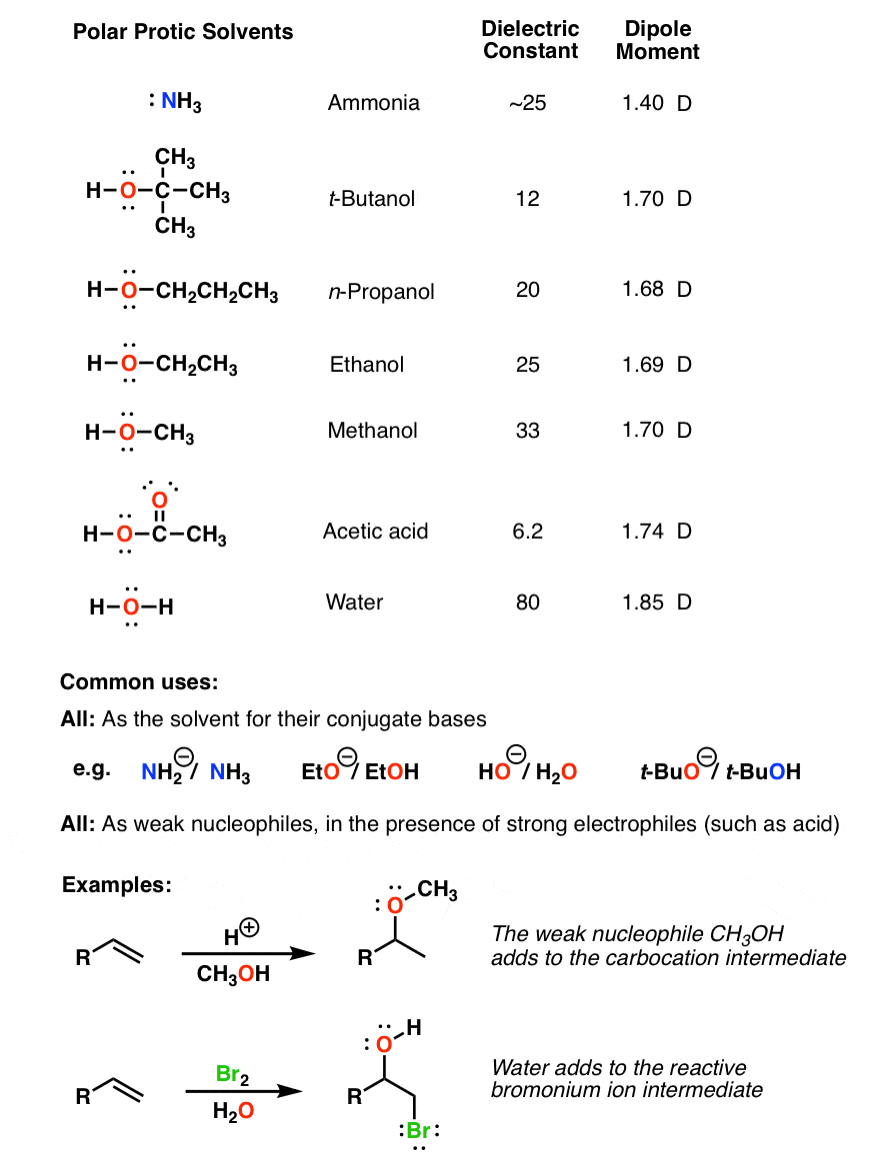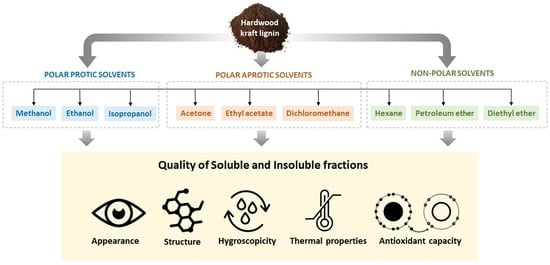
Polymers | Free Full-Text | One-Step Lignin Refining Process: The Influence of the Solvent Nature on the Properties and Quality of Fractions

Organic solvent classification according to the degree of polarity by... | Download Scientific Diagram

What characteristics do methanol, chloroform, diethyl ether, etc. possess that makes them good solvents? - Quora

ick an appropriate solvent(s) from table to dissolve each of the following substances. Common Polar Solvents Common Nonpolar Solvents water (H2O) hexane (C6H12) acetone (CH3COCH3) diethyl ether (CH3CH | Homework.Study.com

Diethyl ether has a much higher boiling point than butane despite having a higher molecular weight. Explain why this is the case, making reference to the molecular structures of both compounds.

Solved: Determine The Solvent Polarity Index For The Follo... | Chegg.com | Index, Solvent, Homework help